|

Lab
Diagnostic Testing: Adenovirus
The laboratory
diagnosis of ocular adenovirus infection is a function of the onset
of clinical symptoms. The earlier the conjunctival samples are collected
after clinical onset, the higher likelihood of a positive result.
The adenoviral load of viable virus and antigen decreases over time.
Specimen Collection
PCR
Specimen
Collection
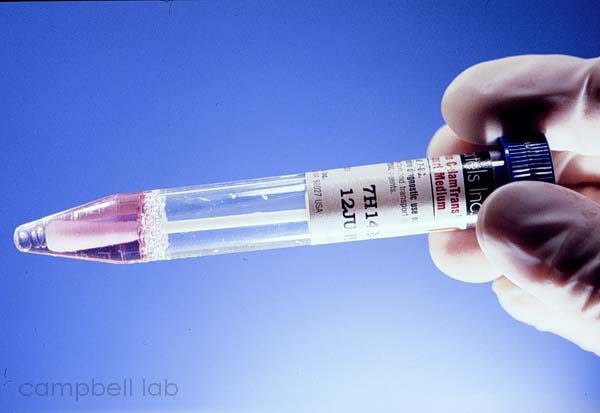
Specimens are directly collected by vigorously swiping the exposed
conjunctiva with a plastic soft-tipped applicator. Cornea samples
are not necessary. Topical anesthetic can be applied to the conjunctiva
but this is optional. Collected samples are placed in 2.0 ml of
viral transport medium. Adenovirus is not a fastidious
virus. It will remain viable under many conditions and collected
samples should be easily transported through mail carriers.
Viral Transport medium
(Click on image to enlarge)
 |
PCR
Polymerase Chain Reaction (PCR) is a molecular test that amplifies
specific adenoviral DNA sequences from clinical samples and then
identifies the amplified products with gel techniques. PCR is a
highly sensitive and specific test that can detect adenoviral DNA
from clinical samples. Results can now be obtained within one to
three days. PCR testing can be quite expensive
to the self-pay patient. Make sure the patient has insurance coverage.
Many molecular laboratories offer adenoviral PCR testing.

|

